AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog

In addition, every α-carbon is attached to one hydrogen, where the R group and the hydrogen always have the same arrangement with respect to the rest of the amino acid.įor example, an amino acid is shown below. The sidechains are the “R” groups from the amino acid structure on the previous page and they branch off of the α-carbon. 
The image below highlights the backbone of a sequence of amino acids. The backbone consists of the amide bond (or amide forming amine and carboxyl) as well as the carbon in between each amide, referred to as the α-carbon. The Diversity of Amino AcidsĪmino acids are often considered in two portions: the backbone and the sidechains. Proteins are formed from long, linear polymers of covalently bonded amino acids arranged in a particular sequence as dictated by the genetic code for that protein. 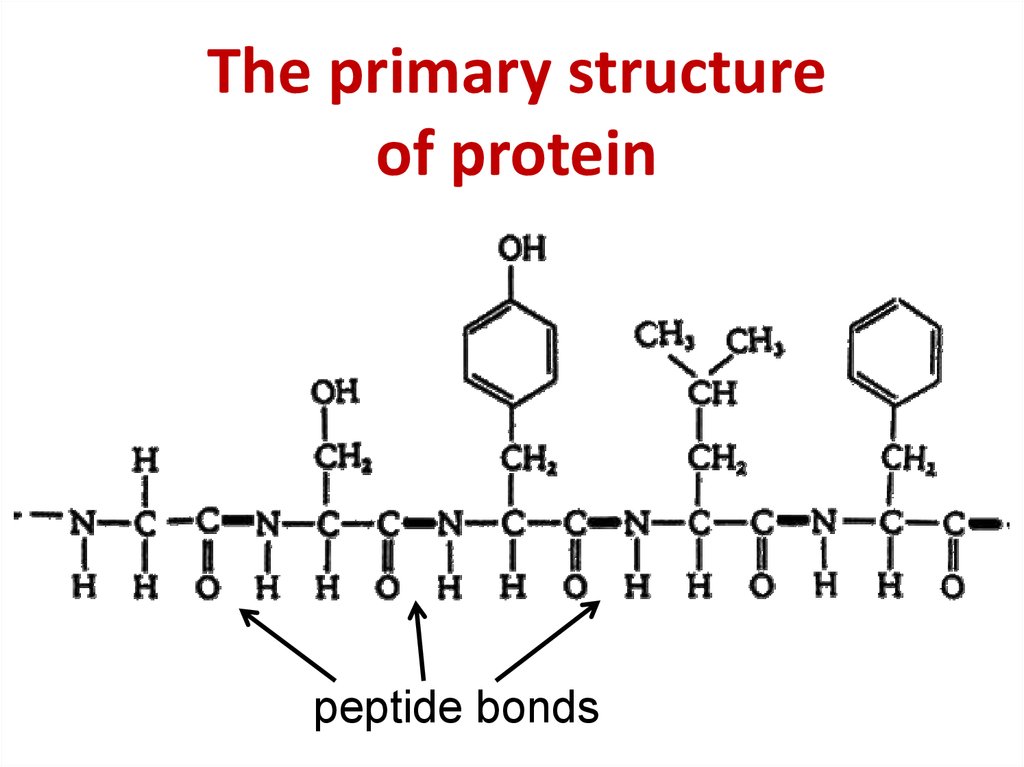
This occurs with the loss of water (an –OH from the carboxyl and an –H from the amine) and is known as dehydration synthesis. The carboxylic acid group (-COOH) is acidic and will have lost its proton to the water, so is shown here as its conjugate base, the carboxylate (-COO –).Ī protein is assembled by forming covalent bonds between amine nitrogens and carbonyl carbons to form amide bonds, aka peptide bonds. The amine group (-NH 2) is basic and will accept a proton from water, so is shown here in its protonated, conjugate acid form (-NH 3 +). 
This structure is shown in the physiologically relevant form, assuming a near-neutral pH aqueous environment. In the amino acid shown at the right, the amino group is shown in blue and the acid group is shown in red. The building blocks are called amino acids. For a structural protein, the dimensions and rigidity of the structure must be suited to its function.Ĭonsidering this complexity, proteins are made up of combinations of (usually) only 20 building blocks comprised of (usually) only 5 different elements. For a membrane protein, the protein must expose a hydrophobic core that will align with the hydrophobic membrane it resides in. For an enzyme, the protein must recognize and bind to a substrate molecule in a way that is favorable to the desired chemical reaction. The shape of a protein is essential to its function.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
